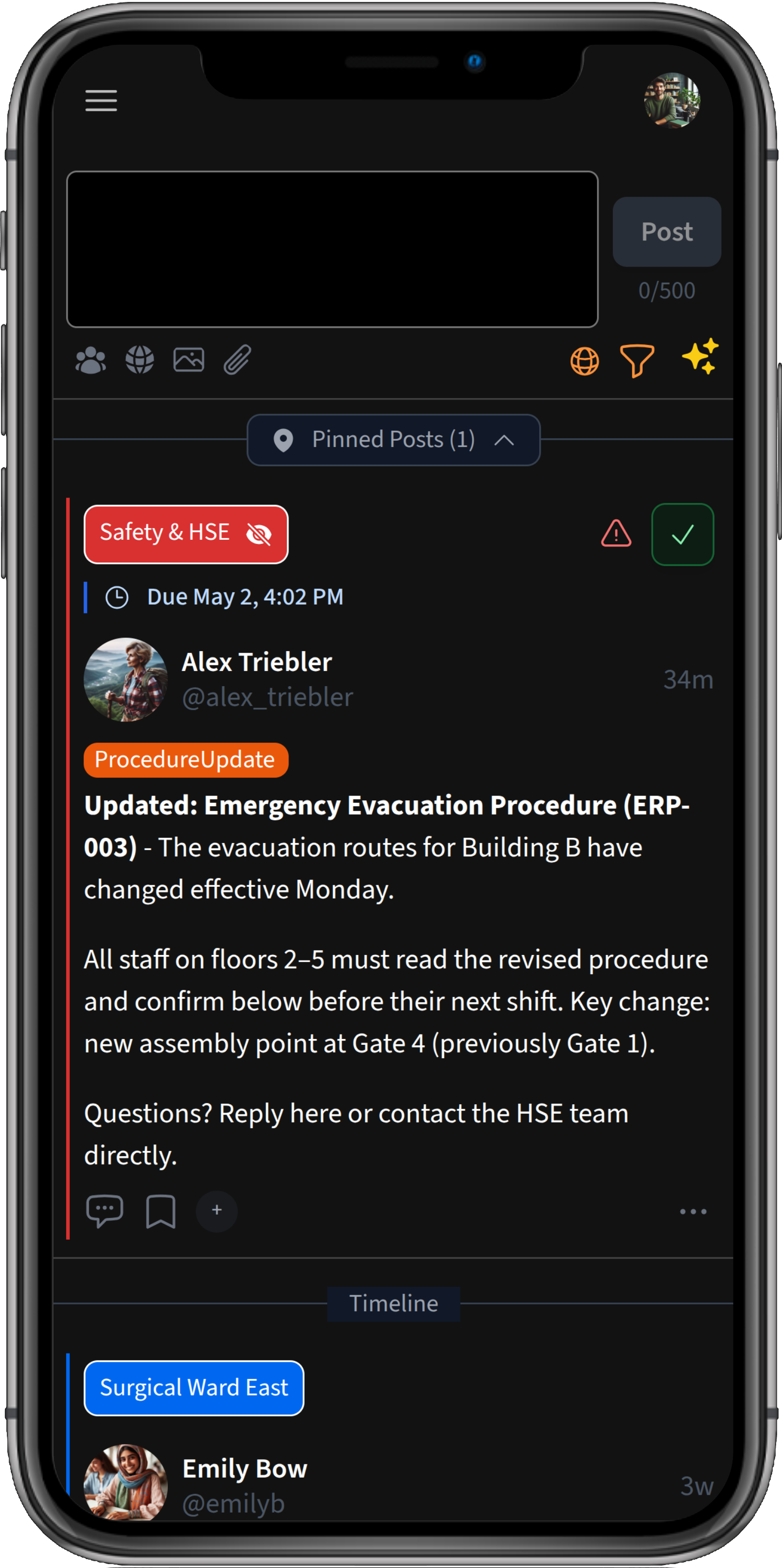
Use case
Know who has confirmed every critical procedure change
When SOPs, safety instructions, or compliance guidelines change, Chirm notifies the right people, requires them to confirm they have understood the update, and lets you follow up with anyone who hasn't.
When a procedure changes — whether it's a safety instruction, a work process, or a compliance requirement — sending an email is not enough. You need to know who has actually read and understood the update, and be able to follow up with those who haven't.
Chirm's critical posts and acknowledgment features give teams a structured way to communicate procedure changes, confirm they've been understood, and keep a clear record, without manual tracking or chasing individuals.
Good fit for
Who benefits
Quality managers, HSE leads, operations managers, and compliance teams
Problem: Procedure changes get sent but not confirmed
A safety instruction changes. A new SOP is issued. A compliance requirement is updated.
The document is finalized, the email goes out — and then nothing. No way to know who opened it, who read it, who understood it. Follow-up happens through individual messages or manual sign-off sheets that are slow, inconsistent, and hard to audit.
The gap is not in the content of the update. The gap is in the confirmation.
"Did everyone across the team actually see the new procedure?"
Without a systematic answer, the risk stays open.
How this works with Chirm
- 1
The responsible team publishes the procedure change in Chirm, targeting the relevant group or location
- 2
The author marks the post as requiring acknowledgment, and optionally as critical; recipients must explicitly confirm they have read and understood it
- 3
Each team member sees the post in their timeline and acknowledges it with a single tap or click
- 4
The responsible person tracks acknowledgment status in real time: who has confirmed, who is still pending
- 5
Automated or manual reminders can be sent to those who have not yet acknowledged
- 6
The full acknowledgment record is stored and can be referenced for audits or inspections
Before and after
| Without Chirm | With Chirm | |
|---|---|---|
| Procedure update delivery | Email or printed sheet, no tracking | Targeted critical post to the right group |
| Confirmation of receipt | Manual sign-off or assumed | Digital acknowledgment per person |
| Follow-up for non-readers | Individual messages, phone calls | Visible pending list with direct follow-up |
| Audit evidence | Paper files or email threads | Searchable acknowledgment record in Chirm |
| Confidence level | Low, based on assumptions | High, based on confirmed data |
Who this is relevant for inside the organization
Quality and compliance teams
Publish SOP changes and policy updates with confidence that the right people have confirmed receipt, backed by an audit trail.
HSE and safety managers
Ensure safety-critical procedure changes reach every relevant employee before work continues. Track who is still pending without manual follow-up.
Operations and team leads
See at a glance which team members have acknowledged a new instruction and follow up directly with those who haven't, without leaving Chirm.
HR and people operations
Communicate policy changes, handbook updates, or compliance requirements and maintain a clear record of who has been informed.
Relevant Chirm features
- Required acknowledgment: mark posts so recipients must explicitly confirm they have read and understood them
- Critical posts: optionally flag a post as critical for additional urgency
- Acknowledgment tracking: see who has confirmed and who is still pending
- Groups and topics: target the exact team, location, or role affected
- Reminders: follow up with non-readers without manual outreach
- Audit record: searchable log of who acknowledged what and when
- Timeline: procedure updates appear alongside regular communication, in context
Example KPIs for a 90-day pilot
| KPI | Observation question | What progress looks like |
|---|---|---|
| Acknowledgment rate | What share of targeted recipients confirm critical posts within the expected timeframe? | Acknowledgment rate above 90% within agreed SLA |
| Time to full acknowledgment | How long does it take from publication until all required recipients have confirmed? | Measurable reduction compared to previous email or paper process |
| Follow-up effort | How much manual effort is needed to follow up with recipients who haven't confirmed after a procedure change? | Significant reduction in individual follow-up messages or calls |
| Audit readiness | Can the team produce a complete acknowledgment record for a given procedure change on demand? | Full record available within minutes, without manual compilation |
4-week rollout plan
- 1
Week 1: Identify the two to three procedure types that most need reliable confirmation: typically safety instructions, compliance updates, or recurring SOPs
- 2
Week 2: Publish the first critical posts for upcoming procedure changes and brief the relevant teams on how acknowledgment works
- 3
Week 3: Review acknowledgment rates, identify where follow-up is still needed, and collect feedback from team leads
- 4
Week 4: Share results with stakeholders, document the first complete acknowledgment records, and extend to additional procedure types
FAQ for rollout teams
Is Chirm a document management system?
No. Chirm does not replace your existing document or SOP management tools. It handles the communication and confirmation step: making sure the right people see a change, acknowledge it, and that the confirmation is recorded.
What happens if someone misses the critical post?
The post remains visible in the recipient's timeline until they acknowledge it. Responsible parties can see who is still pending and send targeted reminders without leaving Chirm.
Can we limit critical posts to specific teams or locations?
Yes. Groups and topics in Chirm let you target exactly the people affected by a procedure change (a specific department, shift, location, or role) without sending irrelevant notifications to everyone.
How is this different from just sending an email?
Email has no reliable read confirmation and no structured follow-up. Chirm records each acknowledgment individually, shows who is still pending in real time, and keeps the confirmation log searchable for future reference.
Is the acknowledgment record suitable for audits?
Chirm stores who acknowledged a critical post and when. Whether that record meets specific regulatory audit requirements depends on your industry and jurisdiction; speak to your compliance team about how it fits your specific framework.
Can I directly add files to the post?
Yes. Chirm supports adding attachments, images, and links directly to a post. This makes it easy to include the updated SOP document, a revised procedure PDF, or a link to further documentation right alongside the notification.
Do employees need training to use it?
No. Acknowledging a critical post is a single action, a tap or click. Most employees are up and running within minutes.